Alzheimer’s-in-a-dish is ‘first clear evidence’ for amyloid hypothesis
October 13, 2014
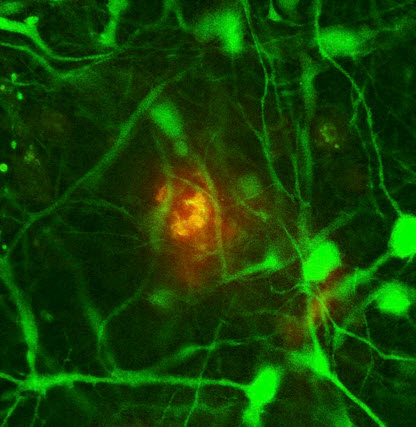
A confocal microscope image of an amyloid-beta deposit (red-orange) in 3D neural cell culture (credit: Se Hoon Choi et al./Nature)
Massachusetts General Hospital (MGH) researchers have created the first “Alzheimer’s-in-a-dish” — a 3D petri dish capable of reproducing the full course of events underlying the development of Alzheimer’s disease.
Alzheimer’s has been thought to result from the buildup of inflammatory plaque formed by the beta-amyloid protein and from another protein, tau, which entangles neurons.
The new research provides the first clear evidence supporting the hypothesis that deposition of beta-amyloid plaques in the brain is in fact the first step in a cascade leading to the devastating neurodegenerative disease, the researchers say. The research also identified the essential role in that process of a specific enzyme that could be a therapeutic target.
Failure of 2D research methods
“Originally put forth in the mid-1980s, the amyloid hypothesis maintained that beta-amyloid deposits in the brain set off all subsequent events — the neurofibrillary tangles that choke the insides of neurons, neuronal cell death, and inflammation leading to a vicious cycle of massive cell death,” says Rudolph Tanzi, PhD, director of the MGH Genetics and Aging Research Unit and co-senior author of the report in Nature.
Previous research was inconclusive, the researchers say. The mouse models of Alzheimer’s disease developed either amyloid plaques or neurofibrillary tangles, but not both. And cultured neurons from human patients with Alzheimer’s exhibited elevated levels of the toxic form of amyloid found in plaques and the abnormal version of the tau protein that makes up tangles, but not actual plaques and tangles.
Genetics and Aging Research Unit investigator Doo Yeon Kim, PhD, co-senior author of the Nature paper, realized the problem: the liquid two-dimensional systems usually used to grow cultured cells poorly represent the gelatinous three-dimensional environment within the brain. So the MGH team developed a gel-based, 3D culture system to grow human neural stem cells that carried variants in two genes — the amyloid precursor protein and presenilin 1 — known to underlie early-onset familial Alzheimer’s Disease (FAD). Both of those genes were previously discovered in Tanzi’s laboratory.
Revolutionizing drug discovery for neurodegenerative disorders
After growing for six weeks, the FAD-variant cells were found to have significant increases in both the typical form of beta-amyloid and the toxic form associated with Alzheimer’s. The variant cells also contained the neurofibrillary tangles that choke the inside of nerve cells causing cell death.
In addition, blocking steps known to be essential for the formation of amyloid plaques prevented the formation of both the tangles (confirming amyloid’s role in initiating the process), and inhibiting the action of an enzyme called GSK3-beta prevented the formation of tau aggregates and tangles*.
“This new testing system — which can be adapted to other neurodegenerative disorders — should revolutionize drug discovery in terms of speed, costs and physiologic relevance to disease,” says Tanzi. “Testing drugs in mouse models that typically have brain deposits of either plaques or tangles, but not both, takes more than a year and is very costly.
“With our three-dimensional model that recapitulates both plaques and tangles, we now can screen hundreds of thousands of drugs in a matter of months without using animals in a system that is considerably more relevant to the events occurring in the brains of Alzheimer’s patients.”
Tanzi is the Kennedy Professor of Child Neurology and Mental Retardation, and Kim is an assistant professor of Neurology at Harvard Medical School. The study was supported by a grant from the Cure Alzheimer’s Fund and by two National Institute of Health grants. MGH is the original and largest teaching hospital of Harvard Medical School.
* The version of tau found in tangles is characterized by the presence of excess phosphate molecules, and when the team investigated possible ways of blocking tau production, they found that inhibiting the action of an enzyme called GSK3-beta — known to phosphorylate tau in human neurons — prevented the formation of tau aggregates and tangles even in the presence of abundant beta-amyloid and amyloid plaques.
Abstract of Nature paper
Alzheimer’s disease is the most common form of dementia, characterized by two pathological hallmarks: amyloid-b plaques and neurofibrillary tangles. The amyloid hypothesis of Alzheimer’s disease posits that the excessive accumulation of amyloid-b peptide leads to neurofibrillary tangles composed of aggregated hyperphosphorylated tau. However, to date, no single disease model has serially linked these two pathological events using human neuronal cells. Mouse models with familial Alzheimer’s disease (FAD) mutations exhibit amyloid-b-induced synaptic and memory deficits but they do not fully recapitulate other key pathological events ofAlzheimer’s disease, including distinct neurofibrillary tangle pathology. Human neurons derived from Alzheimer’s disease patients have shown elevated levels of toxic amyloid-b species and phosphorylated tau but did not demonstrate amyloid-b plaques or neurofibrillary tangles. Here we report that FAD mutations in b-amyloid precursor protein and presenilin 1 are able to induce robust extracellular deposition of amyloid-b, including amyloid-b plaques, in a human neural stem-cell-derived three-dimensional (3D) culture system. More importantly, the 3D differentiated neuronal cells expressing FAD mutations exhibited high levels of detergent-resistant, silver-positive aggregates of phosphorylated tau in the soma and neurites, as well as filamentous tau, as detected by immunoelectron microscopy. Inhibition of amyloid beta generation with b- or c-secretase inhibitors not only decreased amyloid-b pathology, but also attenuated tauopathy.We also found that glycogen synthase kinase 3 regulated amyloid-b-mediated tau phosphorylation. We have successfully recapitulated amyloid-beta and tau pathology in a single 3D human neural cell culture system. Our unique strategy for recapitulating Alzheimer’s disease pathology in a 3D neural cell culture model should also serve to facilitate the development of more precise human neural cell models of other neurodegenerative disorders.
