Intracellular microlasers for precise labeling of a trillion individual cells
August 3, 2015

Massachusetts General Hospital investigators have induced subcutaneous fat cells in a piece of skin from a pig to emit laser light in response to energy delivered through an optical fiber (credit: Matjaž Humar and Seok Hyun Yun/Nature Photonics)
Imagine being able to label a trillion cells in the body to detect what’s going on in each individual cell.
That’s the eventual goal of a Massachusetts General Hospital (MGH) study to allow individual cells to produce laser light. The wavelengths of light emitted by these intracellular microlasers differ based on factors such as the size, shape, and composition of each microlaser, allowing precise labeling of individual cells.
“The fluorescent dyes currently used for research and for medical diagnosis are limited because they emit a very broad spectrum of light,” explains Seok Hyun Yun, PhD, of the Wellman Center for Photomedicine at MGH, corresponding author of the report. “As a result, only a handful of dyes can be used at a time, since their spectral signatures would overlap.”
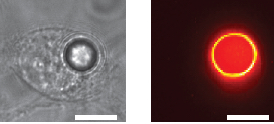
(Left) Bright-field image of a HeLa cell containing a polystyrene fluorescent bead. (Right) False-color image of the cell. (scale bars: 10 micrometers) (credit: Matjaž Humar and Seok Hyun Yun/Nature Photonics)
Lead author Matjaž Humar, PhD, also of the Wellman Center, adds, “The narrow-band spectrum of light emitted by these intracellular lasers would allow us to label thousands — in principle, up to a trillion — of cells individually [the estimated number of cells in the human body], and the very specific wavelengths emitted by these microlasers also would allow us to measure small changes happening within a cell with much greater sensitivity than is possible with broadband fluorescence.”
The trick is to use solid plastic fluorescent microbeads, which are readily taken up into cells, each with a unique signature spectrum based on the size and number of beads within a cell and the fluorescent dye used.
“One immediate application of these intracellular lasers could be basic studies, such as understanding how cells move and respond to external forces,” says Yun, an associate professor of Dermatology at Harvard Medical School.
“Another challenging step will be figuring out how to use biologically generated energy from mechanical movement or a biochemical reaction to pump a cellular laser in a living body. Cells are smart machines, and we are interested in exploiting their amazing capabilities by developing smart-cell lasers that might be able to find diseases and fire light at them on their own.
“We can envision lasers completely made out of materials that are safe for use within the human body, which could enable remote sensing within the body or be used in laser-light therapies.”
The researchers’ report has received Advance Online Publication in Nature Photonics.
Abstract of Intracellular microlasers
Optical microresonators, which confine light within a small cavity, are widely exploited for various applications ranging from the realization of lasers and nonlinear devices to biochemical and optomechanical sensing. Here we use microresonators and suitable optical gain materials inside biological cells to demonstrate various optical functions in vitro including lasing. We explore two distinct types of microresonator—soft and hard—that support whispering-gallery modes. Soft droplets formed by injecting oil or using natural lipid droplets support intracellular laser action. The laser spectra from oil-droplet microlasers can chart cytoplasmic internal stress (∼500 pN μm–2) and its dynamic fluctuations at a sensitivity of 20 pN μm–2 (20 Pa). In a second form, whispering-gallery modes within phagocytized polystyrene beads of different sizes enable individual tagging of thousands of cells easily and, in principle, a much larger number by multiplexing with different dyes.
