Mice with MS-like condition walk again after neural stem-cell treatment
May 19, 2014
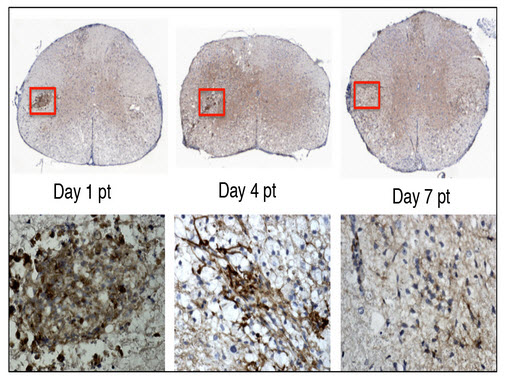
(Top row) Stem cells transplanted into the mouse spinal cord. (Bottom row) Close up of the stem cells (brown). By Day 7 post transplant (pt), the stem cells are no longer detectable. They have sent chemical signals to the mouse¹s own cells, enabling them to repair the nerve damage similar to that seen in multiple sclerosis. (Credit: Lu Chen)
When scientists transplanted human neural stem cells into mice with multiple sclerosis (MS), within a remarkably short period of time, 10 to 14 days, the mice had regained motor skills.
Six months later, they showed no signs of slowing down.
Results from the study demonstrate that the mice experience at least a partial reversal of symptoms. Immune attacks are blunted, and the damaged myelin is repaired, explaining their dramatic recovery.
The finding, which uncovers potential new avenues for treating MS, was published May 15, 2014 in the journal Stem Cell Reports (open access).
How they did it
Ronald Coleman (a graduate student of Jeanne Loring, Ph.D., co-senior author and director of the Center for Regenerative Medicine at The Scripps Research Institute and co-first author on the publication) changed the normal protocol and grew the neural stem cells so they were less crowded on a Petri dish than usual.
That yielded a human neural stem cell type that turned out to be extremely potent. The experiments have since been successfully repeated with cells produced under the same conditions, but by different laboratories.
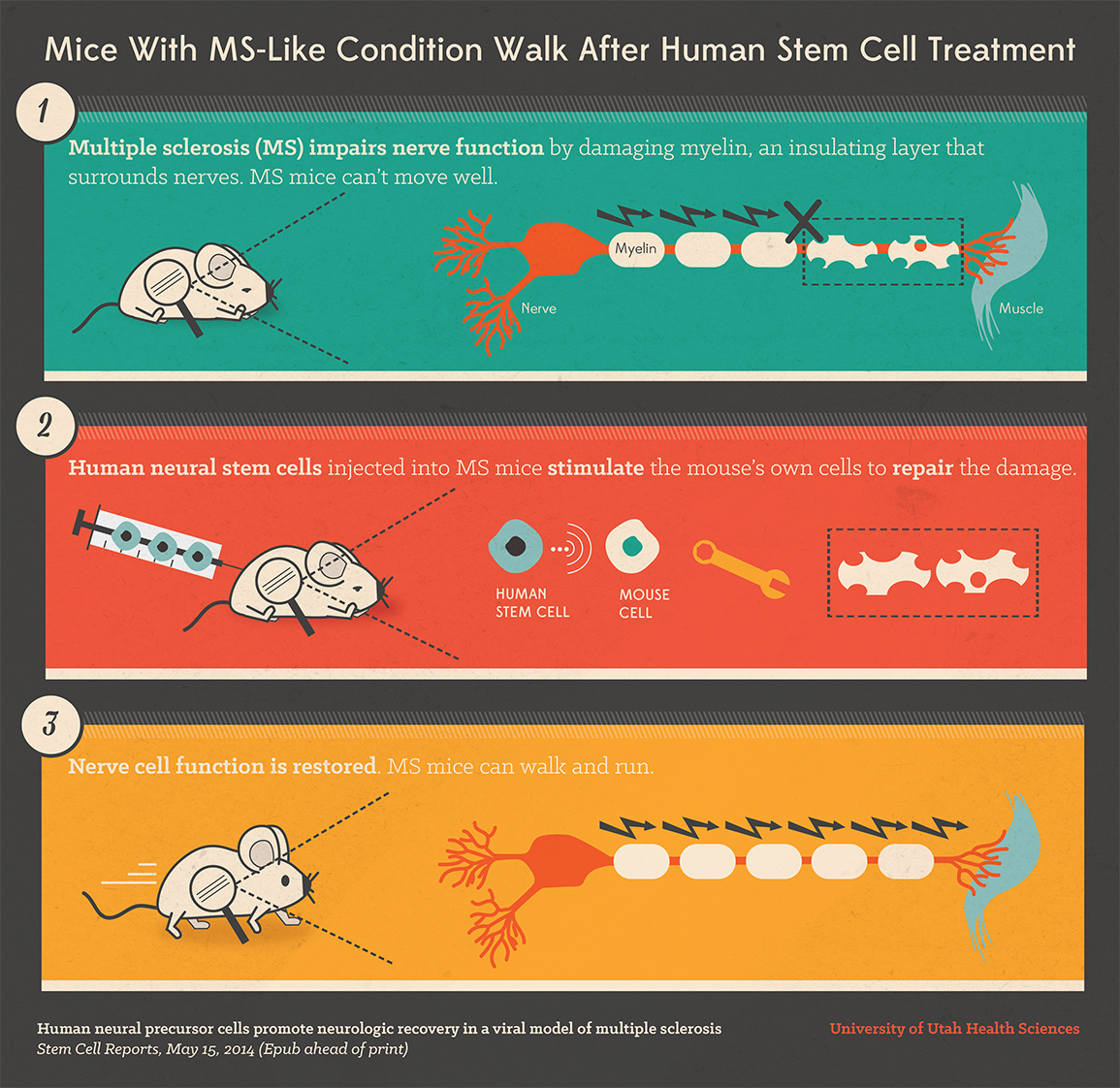
1) Multiple sclerosis (MS) impairs nerve function by damaging myelin, an insulating layer that surrounds nerves. MS mice can’t move well. 2) Human neural stem cells injected into MS mice stimulate the mouse’s own cells to repair the damage. 3) Nerve cell function is restored. MS mice can walk and run. (Credit: University of Utah Health Sciences Office of Public Affairs)
The human neural stem cells send chemical signals that instruct the mouse’s own cells to repair the damage caused by MS. Experiments by Lane’s team suggest that TGF-beta proteins comprise one type of signal, but there are likely others. This realization has important implications for translating the work to clinical trials in the future.
“Rather than having to engraft stem cells into a patient, which can be challenging from a medical standpoint, we might be able to develop a drug that can be used to deliver the therapy much more easily,” said Tom Lane, Ph.D., a professor of pathology at the University of Utah.
With clinical trials as the long-term goal, the next steps are to assess the durability and safety of the stem cell therapy in mice. “We want to try to move as quickly and carefully as possible,” he said. “I would love to see something that could promote repair and ease the burden that patients with MS have.”
“The aspect I am most interested is to define what is being secreted from the human cells that influence demyelination,” Lane told KurzweilAI in an email interview. “Other studies have shown either effects on neuroinflammation or demyelination; ours is one of a select few to show that stem cells influence both.”
However, it is too soon to say when can we expect this innovation to be available for MS patients, Lane added.
UC Irvine and University of Colorado, Aurora researchers were also involved in the research, which was funded by the National Multiple Sclerosis Society and the California Institute of Regenerative Medicine.
Background
More than 2.3 million people worldwide have MS, a disease in which the immune system attacks myelin, an insulation layer surrounding nerve fibers. The resulting damage inhibits transmission of nerve impulses, producing a wide array of symptoms including difficulty walking, impaired vision, fatigue and pain.
Current FDA-approved medications slow early forms of the disease by dampening attacks by the immune system. In recent years, scientists have turned their attention to searching for ways to halt or reverse MS. Such a discovery could help patients with latter, or progressive, stages of the disease, for whom there are no treatments.
Abstract of Stem Cell Reports paper
- Spinal cord transplantation of hNPCs results in recovery in a viral model of MS
- hNPC-mediated recovery occurs in the absence of engrafted cells
- hNPCs are immunomodulatory through increasing the frequency of Tregs in the CNS
- hNPCs increase Treg frequency via a TGF-β1- and TGF-β2-dependent pathway
Using a viral model of the demyelinating disease multiple sclerosis (MS), we show that intraspinal transplantation of human embryonic stem cell-derived neural precursor cells (hNPCs) results in sustained clinical recovery, although hNPCs were not detectable beyond day 8 posttransplantation. Improved motor skills were associated with a reduction in neuroinflammation, decreased demyelination, and enhanced remyelination. Evidence indicates that the reduced neuroinflammation is correlated with an increased number of CD4+CD25+FOXP3+ regulatory T cells (Tregs) within the spinal cords. Coculture of hNPCs with activated T cells resulted in reduced T cell proliferation and increased Treg numbers. The hNPCs acted, in part, through secretion of TGF-β1 and TGF-β2. These findings indicate that the transient presence of hNPCs transplanted in an animal model of MS has powerful immunomodulatory effects and mediates recovery. Further investigation of the restorative effects of hNPC transplantation may aid in the development of clinically relevant MS treatments.
